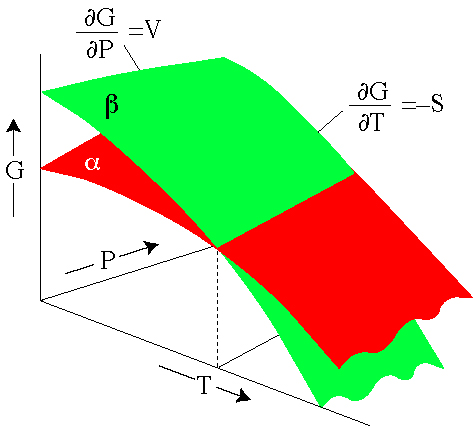
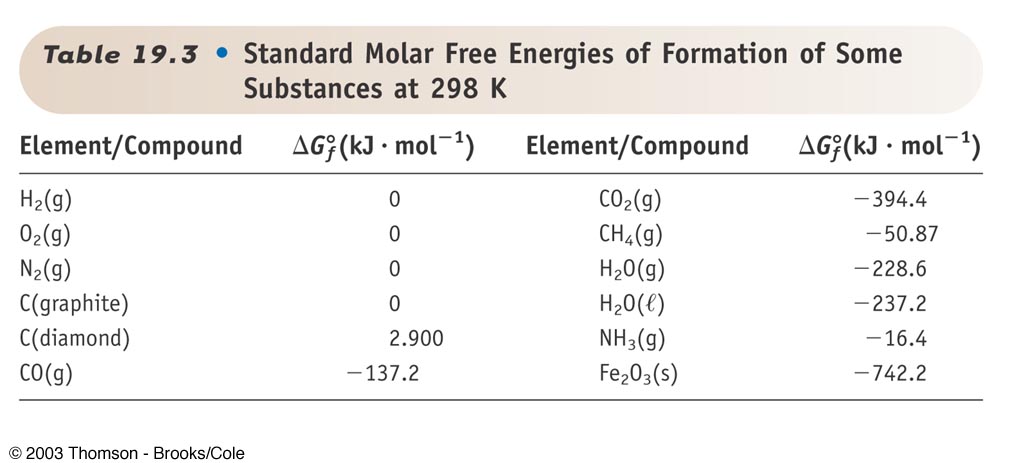
The standard Gibbs free energy of formation of a compound is the change of Gibbs free energy that accompanies the formation of 1 mole of that substance from its component elements at their standard states the most stable form of the element at 25 degrees. The maximum work done is the amount of energy produced given by the decrease in the thermodynamic property called Gibbs free energy.

17 1 Equilibrium And Gibbs Free Energy Hl Youtube
Gibbs free energy equation.

Standard gibbs free energy equation. G H - T D S. ΔH -8904 kJ mol-1. Looking at the following equation we can say if the reaction is reversible and the Gibbs free energy is zero then the system is said to be in equilibrium.
The following equation relates the standard-state free energy of reaction with the free energy at any point in a given reaction not necessarily at standard-state conditions. The free energy change D G is equal to -T D S univ and it applies just to a system itself without regard for the surroundings. You must convert your standard free energy value into.
Determining if a reaction is spontaneous by calculating the change in Gibbs free energy. ΔG ƒ ΔH ƒ compound - TΔS compound Values of standard Gibbs free energy of formation are tabulated for many compounds. Standard Gibbs free energy of formation of a compound can be calculated using standard enthalpy of formation ΔH ƒ absolute standard entropy ΔS and standard temperature T 29815 K.
Standard change in free energy and the equilibrium constant. Fi is the standard-state free energy of formation per mole of species i activity active concentration R the ideal gas constant 1987 cal K-1mol-1 831 J K-1mol T K a b c d A B C D RT. And youll get minus 890 point 3 kilojoules whats tells us that this is an exothermic reaction that this side of.
G H - TS. Free Energy and Free Energy Change the Gibbs free energy G is used to describe the spontaneity of a process. Δ G Δ G ⁰ RT ln Q where R is the ideal gas constant 8314 Jmol K Q is the reaction quotient and T is the temperature in Kelvin.
Hence the standard Gibbs free energy for the transformation of diamond to graphite at 298 K is -28982kJmol. Gibbs Free energy formula is given below. When a system changes from an initial state to a final state the Gibbs free energy ΔG equals the work exchanged by the system with its surroundings minus the work of the pressure force.
Free Energy and the Equilibrium Constant Because ΔH and ΔS determine the magnitude of ΔG and because K is a measure of the ratio of the concentrations of products to the concentrations of reactants we should be able to express K in terms of ΔG and vice versa. D G D H - T D S. ΔH change in enthalpy.
G H - TS If the reaction is run at constant temperature this equation can be written as follows. 129 rows The standard Gibbs free energy of formation G f of a compound is the change of. Gibbs Free Energy Change Concept and Standard Free Energy Change Equation and Sums Which are Very Important for a CSIR NET Examination and other National le.
Become a member and unlock all Study Answers Try it risk-free for 30 days. It is defined by the Gibbs equation. The Gibbs energy for a reaction which is in the standard state ᶿ is related to the equilibrium constant as follows.
So if you had to calculate the Gibbs free energy change at say 298 K you can just slot the numbers in. The change in the Gibbs free energy of the system that occurs during a reaction is therefore equal to the change in the enthalpy of the system minus the change in the product of the temperature times the entropy of the system. ΔG ΔH - TΔS ΔG -8904 - 298-02442 -8176 kJ mol-1.
The Gibbs free energy equation is dependent on pressure. The units of ΔG If you look up or calculate the value of the standard free energy of a reaction you will end up with units of kJ mol-1 but if you look at the units on the right-hand side of the equation they include J - NOT kJ. Delta G Delta Go RT ln Q label110 Delta G free energy at any moment Delta Go standard-state free energy.
Using Standard Change in Gibbs Free Energy Δ G ⁰ The change in Gibbs free energy under nonstandard conditions Δ G can be determined from the standard change in Gibbs free energy Δ G ⁰. Also calculates the change in entropy using table of standard entropies.

1 The Laws Of Thermodynamics In Review 1 The Internal Energy Of An Isolated System Is Constant There Are Only Two Ways To Change Internal Energy Heat Ppt Download

Calculations Of Free Energy And Keq Ck 12 Foundation
Cie Nov 2016 V1 Paper 4 Q3 With Explained Solutions

Gibbs Free Energy Example 2 Numerade

Free Energy Delta G Calculations Pt 7 Youtube

Sandwalk Better Biochemistry The Free Energy Of Atp Hydrolysis

How Does Partial Pressure Affect Gibbs Free Energy Socratic

Gibbs Free Energy Boundless Chemistry

Tang 05 Entropy And Gibb S Free Energy

Answered 6 What Is The Standard Gibbs Free Bartleby

Stoichiometric Equations And Standard Gibbs Free Energy Changes For Download Table

Chapter 17 1 Gibbs Free Energy For A

Free Energy Delta G And Equilibrium Pt 8 Youtube
Solved Calculate The Standard Gibbs Free Energy Change As Chegg Com

How To Calculate Standard Gibbs Free Energy Change

Standard Gibbs Energy An Overview Sciencedirect Topics

Answered Calculate The Standard Change In Gibbs Bartleby

The Relationship Between Free Energy And The Equilibrium Constant Video Lesson Transcript Study Com
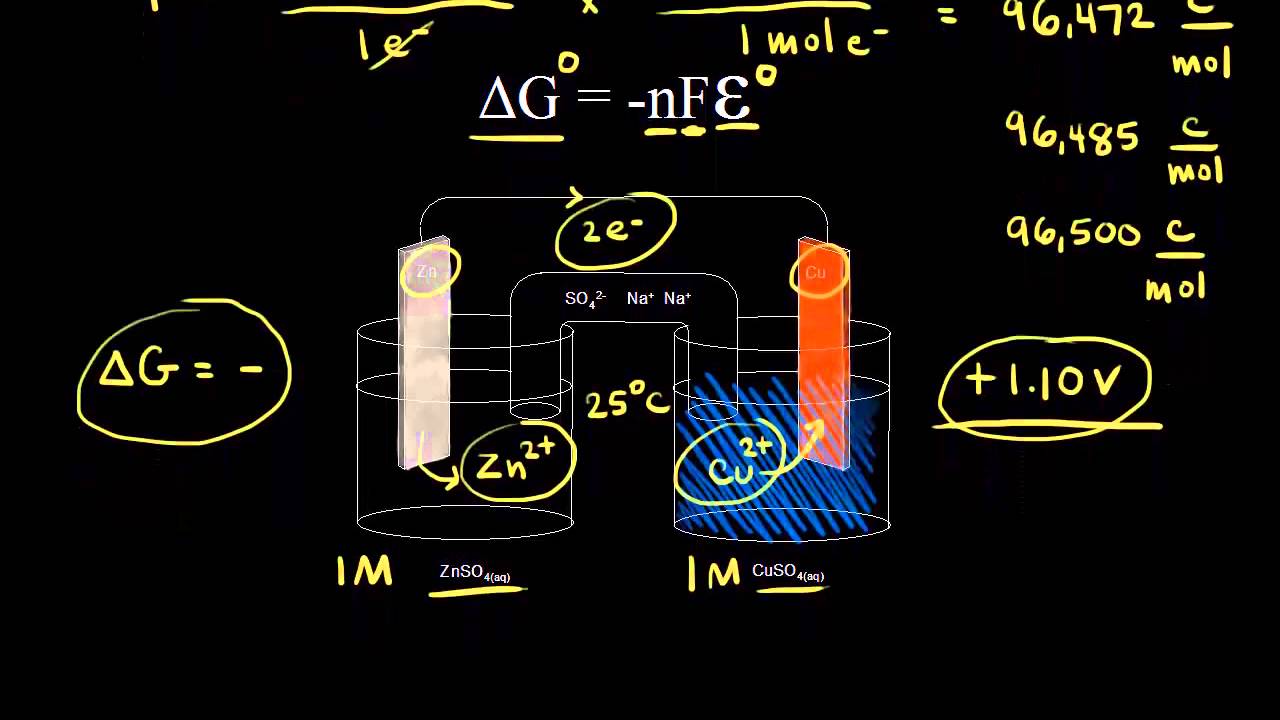
Free Energy And Cell Potential Video Khan Academy

Answered Calculate The Standard Free Energy Of Bartleby

Calculations Of Free Energy And Keq Ck 12 Foundation

Gibbs Free Energy And Problems Stan S Free Energy And Problems 1 Calculate The Standard Gibbs Free Energy Change Associated With The Hydrogenation Of Ethane And Interpret The

Ppt Free Energy And Redox Reactions Powerpoint Presentation Free Download Id 3198993

1 Calculate The Standard Free Energy Change For The Reaction Fe203 S 4h2 8 2fe S 3h20 Brainly In

Topic 15 Energetics Hl Msjchem Tutorial Videos For Ib Chemistry
Temperature In The Gibbs Free Energy Equation Chemistry Stack Exchange

First Law Of Thermodynamics You Will Recall From
Avoiding First Year University Chemistry Textbooks Misrepresentations In The Teaching Of Spontaneous Reactions

Chem 2 Std Free Energy Of Formation Vii

Entropy Free Energy And Equilibrium Chapter 18 Thermodynamics

The Standard Free Energy Change Of A Reaction Is Deltag Kj

Thermodynamics Review The Knowledge You Need To Score High 5 Steps To A 5 Ap Chemistry 2015
How Is Gibbs Free Energy Related To Enthalpy And Entropy Socratic
The Standard Gibbs Free Energy Change G In Kj Mol 1 In A Daniel Cell Ecell 1 1v When 2 Moles Of Zn S Is Oxidized At 298 K Is Closest To

Standard Gibbs Free Energies Of Formation Of Lead And Related Species Download Table

Reaction Quotient And Gibbs Free Energy At The Start Of A Reaction Chemistry Stack Exchange

Difference Between Gibbs Free Energy And Standard Free Energy Compare The Difference Between Similar Terms
Gibbs Free Energy Thermochemistry Training Mcat General Chemistry Review
19 6 Gibbs Energy Change And Equilibrium Chemistry Libretexts

Calculate The Standard Free Energy Of Reaction 1 Youtube

Ap Chemistry Non Standard Gibbs Free Energy Worksheet Review Youtube

17 1 Equilibrium And Gibbs Free Energy Hl Youtube

Temperature In The Gibbs Free Energy Equation Chemistry Stack Exchange

Gibbs Free Energy And Spontaneity And The Meaning

Topic 15 Energetics Hl Msjchem Tutorial Videos For Ib Chemistry
Solved Calculate The Change In Gibbs Free Energy At Stand Chegg Com

Gibbs Free Energy Example Video Khan Academy

Calculate The Standard Change In Gibbs Free Energy Fo
16 4 Gibbs Energy Chemistry Libretexts
Solved 6l 1 Calculate The Standard Reaction Gibbs Free En Chegg Com

Standard Free Energy Changes Introduction To Chemistry

The Relationship Between Free Energy And The Equilibrium Constant Video Lesson Transcript Study Com
Avoiding First Year University Chemistry Textbooks Misrepresentations In The Teaching Of Spontaneous Reactions

What Is The Standard Gibbs Free Energy For This Reaction At 298 K

Standard State Gibbs Free Energy Vs Nonstandard State Gibbs Free Energy Thermodynamics Chemistry Youtube
Ch 4 Gibbs Free Energy Entropy

Chem 2 Std Free Energy Of Formation Vii

Free Energy Chanye Mation Data For The 1 Calculate The Standard Gibbs Free Enery From The Free Energies Of Formation De Following Reaction 15 Ch T 02 9 3

Calculate The Standard Gibbs Free Energy Change For C6h6 15 2 O2 6co2 3h2o Chemistry Thermodynamics 13160574 Meritnation Com

Calculate The Standard Gibbs Free Energy Change From The Free Ener

Calculations Of Henry S Law Constants For Organic Species Using Relative Gibbs Free Energy Change Sciencedirect

Oneclass What Is The Standard Gibbs Free Energy For The Transformation Ofdiamond To Graphite At 298k
Solved Calculate The Standard Gibbs Free Energy For The F Chegg Com

Going Green Free Energy And Equilibrium Equation
Oneclass Ical Reaction The Standard Gibbs Free Energy Of Reaction At 20 0 A C Is 136 Kj Calculate

The Total Pressure Of Reaction Mixture At Equation Is P The Standard Gibbs Free Energy Changeof Brainly In

Chapter 16 Thermodynamics Entropy Free Energy And Equilibrium Ppt Video Online Download

Calculate The Standard Gibbs Free Energy Change Dgo For The Clutch Prep

Gibbs Free Energy Chemistry Video Clutch Prep

Difference Between Enthalpy Entropy Gibbs Free Energy Themodynamics

Gibbs Free Energy How Can We Use H

Getting Gibbs Energy As A Function Of Temperature

Free Energy Definition Biochemistry Going Green
Gibbs Free Energy Equilibrium Thermodynamics

1 Chapter 19 Chemical Thermodynamics Entropy Free Energy And Equilibrium Ppt Download

Cell Potential And Free Energy Protocol
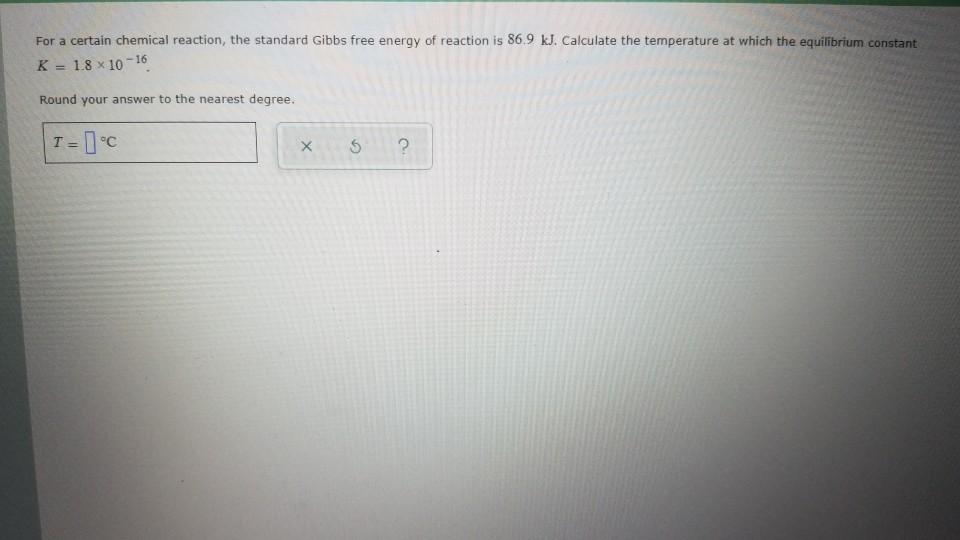
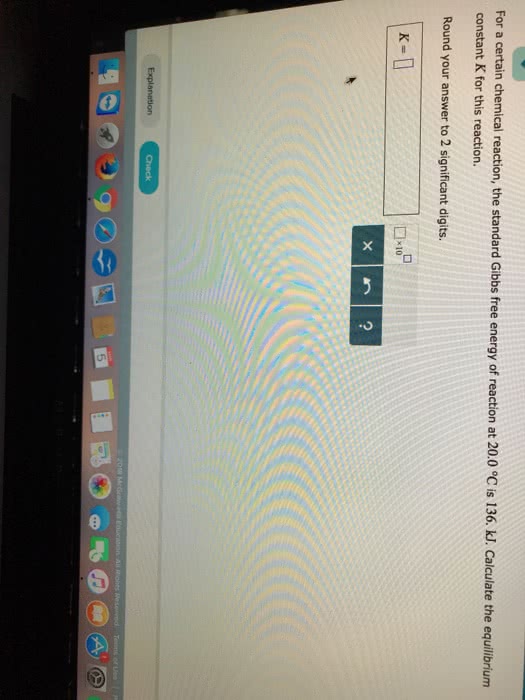
Solved For A Certain Chemical Reaction The Standard Gibb Chegg Com

Apsi 2018 Ap Chemistry 2 Chapter 3 2 By Edvantage Science Issuu
Ppt Gibbs Free Energy Powerpoint Presentation Free Download Id 2198185
First Some Cool Movies Here Is An Animated Form Of The Hcn Cnh Isomerization Click Here For The Isomerization Movie Here Is An Animated Form Of The Imaginary Frequency Of The Transition State Click Here For The Imaginary Frequency Movie Parameter

Calculations Of Free Energy And Keq Ck 12 Foundation

How To Find Q In Gibbs Free Energy
The Standard Gibbs Energy Change At 300 K For The Reaction 2a B C Is 2494 2 J At A Given Time The Composition Of The Reaction Mixture Is A
Chnage Of Standard Gibbs Free Energy For The Decomposition Of Sodium Hydrogen Carbonate Gibbs Free Energy Enthalpy

Calculate The Standard Change In Gibbs Fre Clutch Prep

What Is The Standard Gibbs Free Energy Of Formation Gºf Of Nh3 G At 298 K Brainly Com

The Relationship Between Free Energy And The Equilibrium Constant Video Lesson Transcript Study Com